West Pharmaceutical Services, Inc.
 |
 |
 |
 |
 |
 |
About Us
West is a leading global manufacturer in the design and production of high-quality, technologically advanced containment and delivery systems for injectable medicines. Pharmaceutical, biotechnology, generic and medical device companies trust West and our ability to provide innovative and integrated solutions.
A Commitment to Quality
West's top priority is delivery quality products that meet exact product specifications and quality standards customers require and expect. This focus on quality includes operational excellence, scientific and technical expertise and management. At the manufacturing level, this means producing clean, sterile, high-quality components to minimize disruptions to the supply chain and bringing safe., effective drug and device products to market quickly and efficiently.
Analytical Testing
The pharmaceutical and biotechnical industry are facing challenges as drug development becomes more complex. The regulatory requirements are challenging and resources that understand bringing together both the drug and the device development can be limited.West Analytical Services is experienced not only around injectable components and packaging but devices and combination products as well. Our team has vast expertise and experience in extractables and leachables, particle analysis, container closure integrity, and performance and packaging/delivery systems among other methodologies. As a result of our understanding of materials and delivery systems and their compatibility with the drug product, we can assist our customers in designing studies and in navigating the challenging and evolving regulatory landscape. West Analytical labs are GMP and FDA compliant. The labs are also DEA licensed, certified to ISO 9001:2008 and ISO 15378:2011 and have two MHRA certificates.
Contract Manufacturing
West's Contract Manufacturing group is a process driven, global, contract manufacturer of pharmaceutical and medical devices. With seven locations throughout North America and Europe, the West Contract Manufacturing team is focused on serving the needs of healthcare companies by providing a single sourced solution from product conceptualization through manufacturing and final packaging. Capabilities include: concept design, engineering development, prototyping, production scale up, validation and full scale manufacturing. All of our Contract Manufacturing healthcare facilities are ISO 13485 certified, and cGMP compliant.
- Full-service molding and assembly solutions
- Primary and secondary packaging
- Real-time process monitoring and lean manufacturing principles
- ISO Class 7 and clean room assembly operations (at key facilities)
Our engineers take an innovative yet scientific approach to the development of your products.
- Collaborative cross functional engineering teams
- Rapid component development
- Optimized process for manufacturing efficiencies
- Design for manufacturability (DFM)
Quality Control & Regulatory Expertise
It’s essential to partner with an organization that has established, maintains and applies a compliant quality management system throughout the product life cycle.
- Comprehensive risk management system
- Experienced personnel dedicated to regulatory compliance
- Registered with FDA as a drug repackager/re-labeler(key facilities)
- Seamlessly integrated within manufacturing with independent oversights
- Key Process Indicators (KPI) driven and monitored by plant and by business units
- Validated documented management systems
Check out West Pharma’s latest Blogs:
- Comprehensive Guide to Extractables & Leachables in Pharma Packaging
- Assessing Performance: Novapure Syringe Elastomeric Plungers
- Insights FDA Guidance on Human Factors for Combination Products
- Medical Device Regulations: Annex I Requirements for Staked Needle Syringes
- USP <382>: Fragmentation, Penetration & Sealability of Vial Systems
- USP-382 Packaging Delivery System Integrity and Needle-sealing Capacity of Vial Systems
- USP-382 Drug Packaging Delivery Integrity Needle Self-sealing Capacity Syringe Cartridge Systems
Our Products
Daikyo® Crystal Zenith® Insert Needle Syringe Systems
West Pharmaceutical Services, Inc.The Daikyo® Crystal Zenith® Insert Needle Syringe system...

SelfDose® Patient-Controlled Injector
West Pharmaceutical Services, Inc.The SelfDose® patient-controlled injector is an off-the-shelf delivery system&nbs...
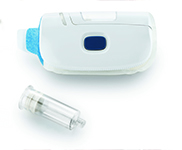
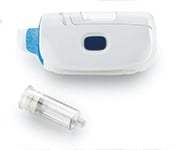
SmartDose® 10 On-Body Delivery System Platform
West Pharmaceutical Services, Inc.SmartDose® 10 On-Body Delivery System. Adaptive technology for large dose...
Contact West Pharmaceutical Services, Inc.
Sign In
Not Signed In
Please sign in or create a free account to request information
Create an AccountNot Signed In
Please sign in or create a free account to save your favorites
Create an Account