In these uncertain times,
Qosina understands it is imperative that its customers quickly adapt to the
changing needs of the medical device industry. Since the beginning of the
COVID-19 crisis, Qosina has been committed to being a one-stop source for
critical medical components.
Qosina
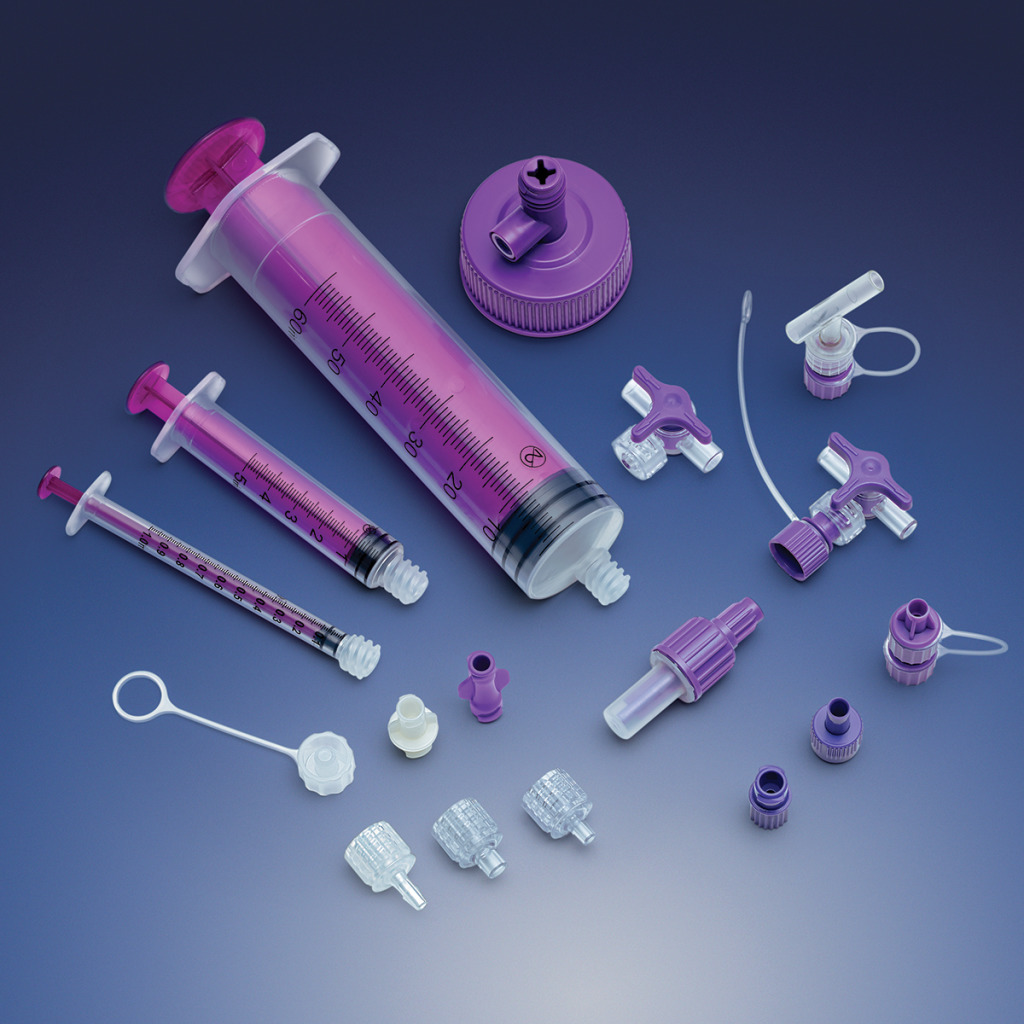
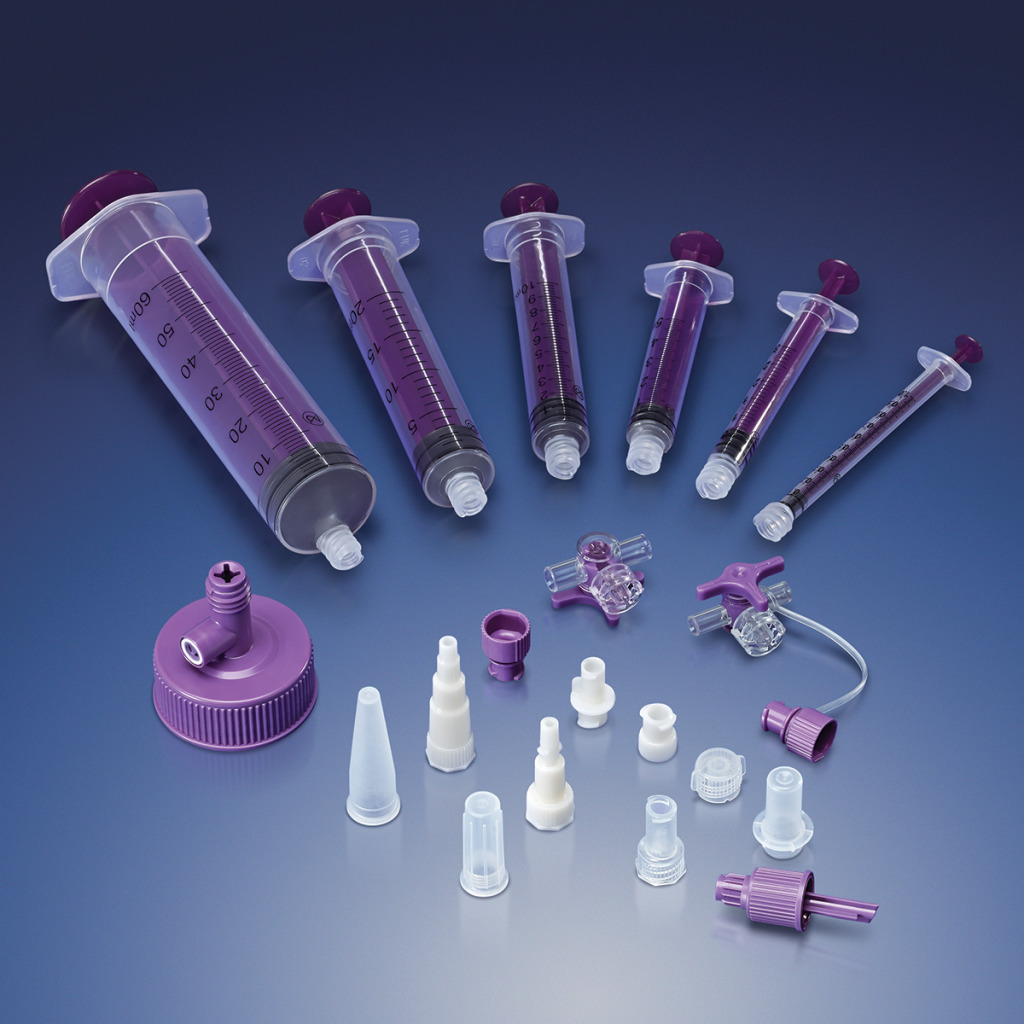
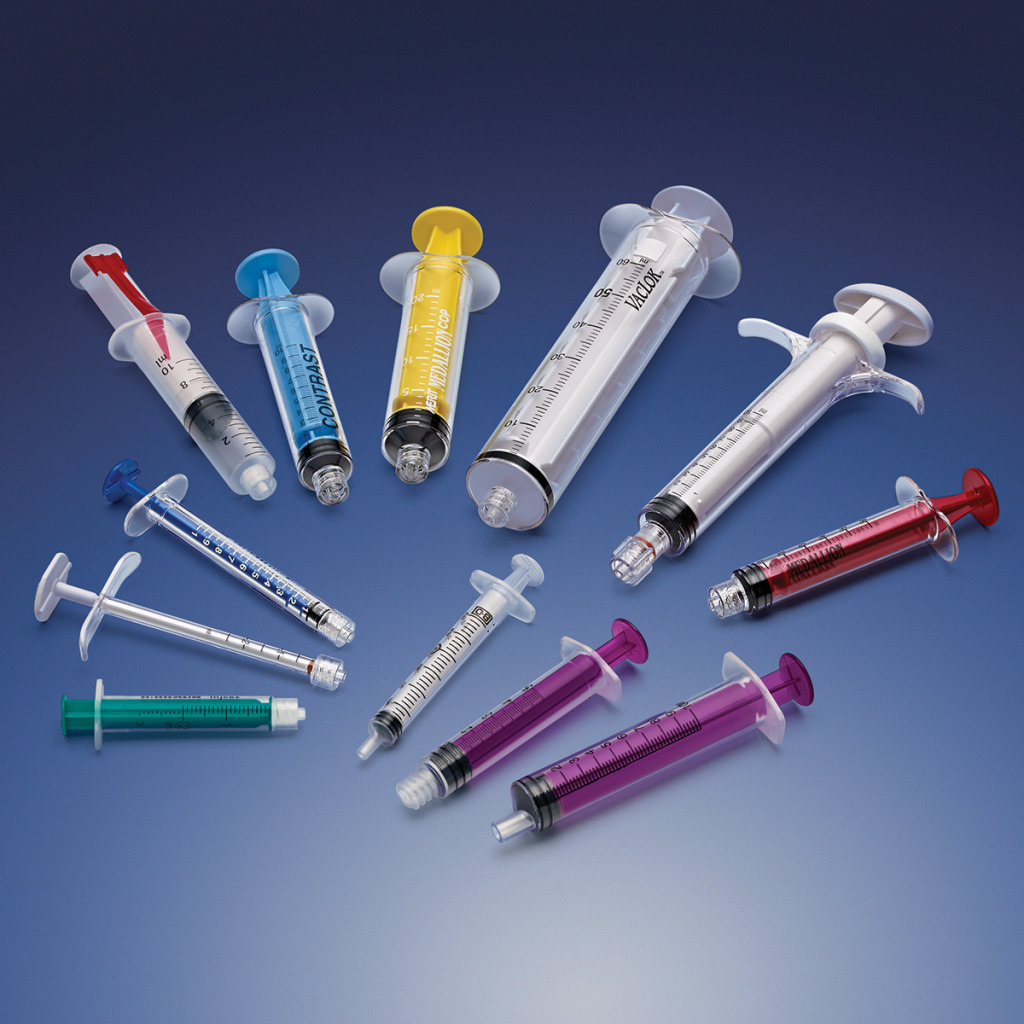
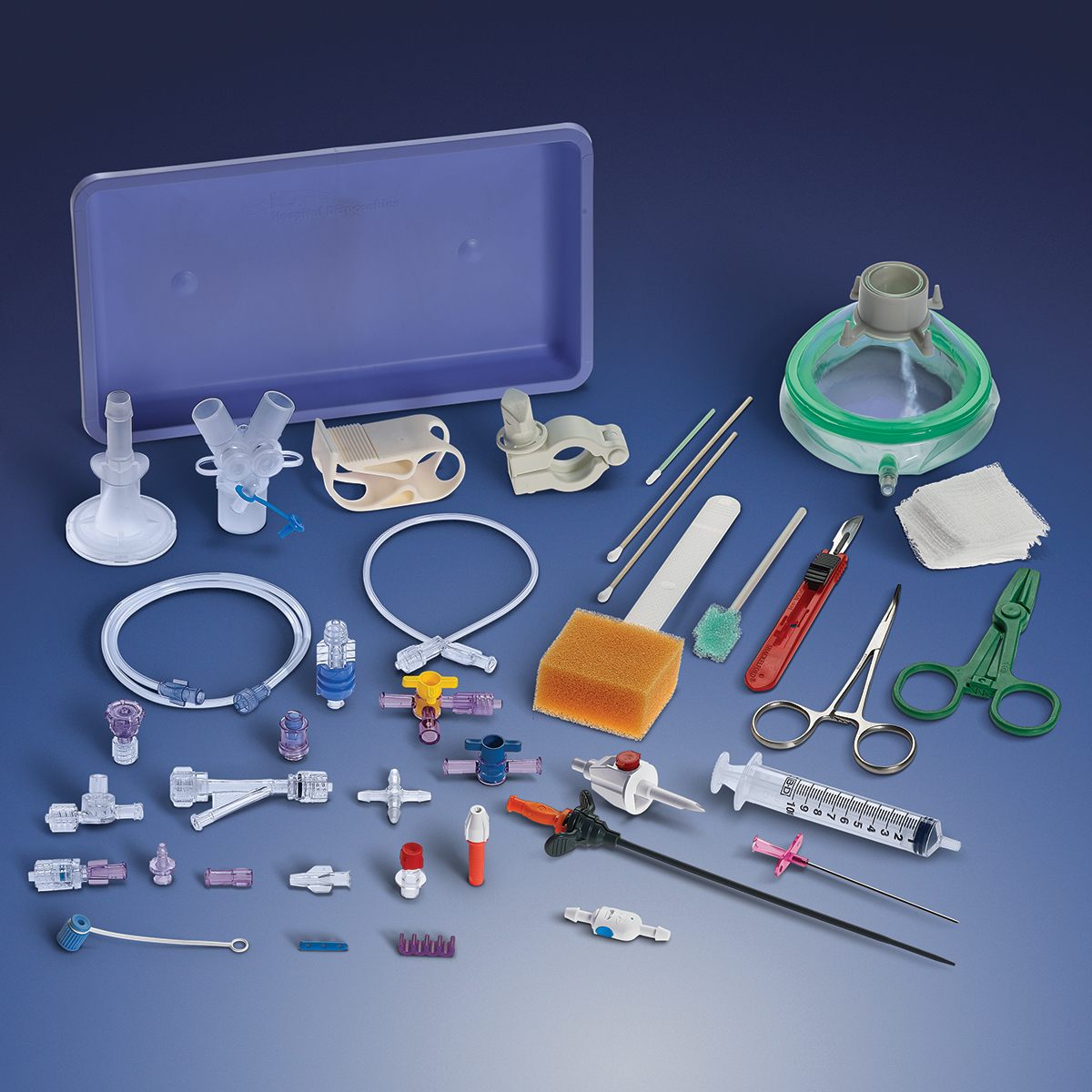
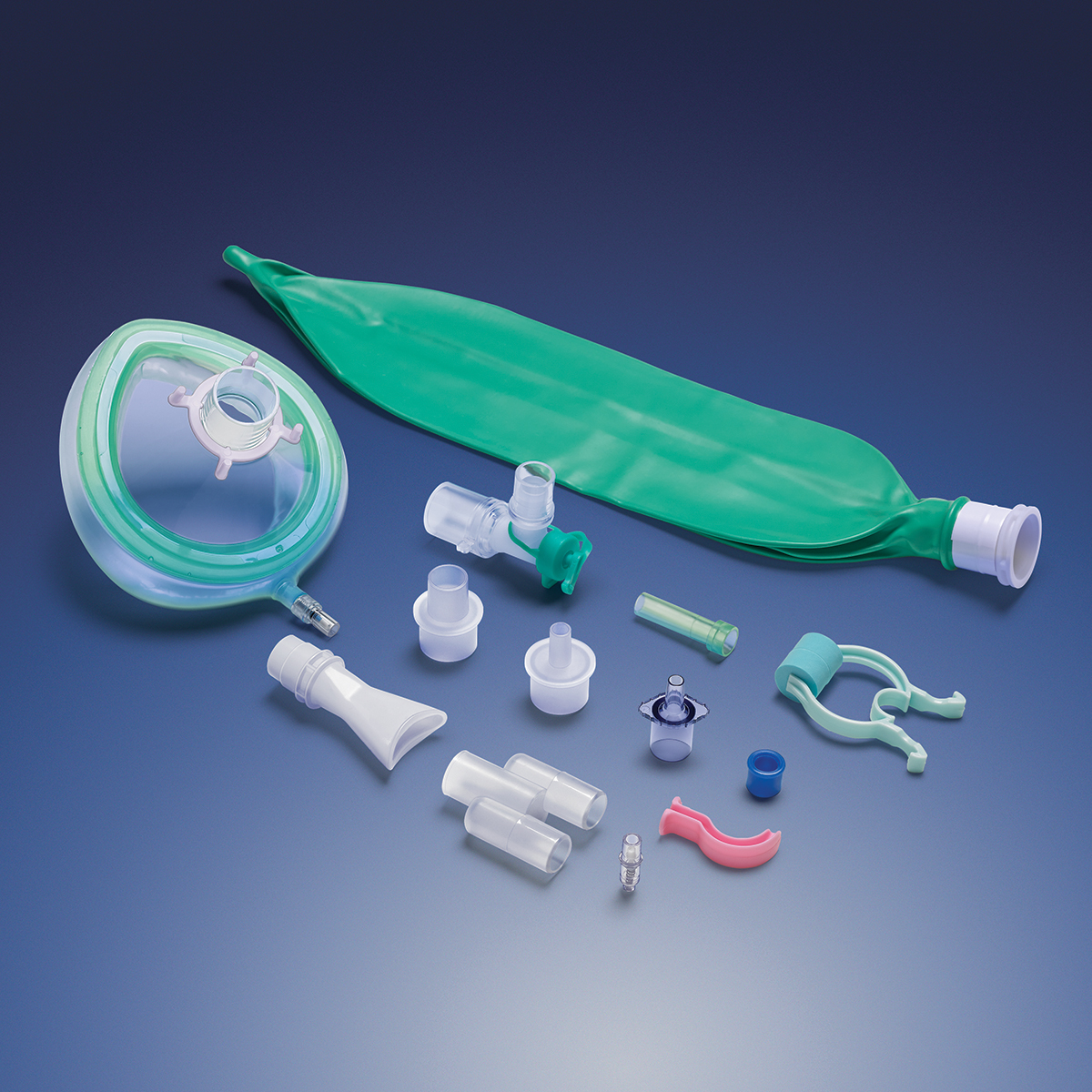
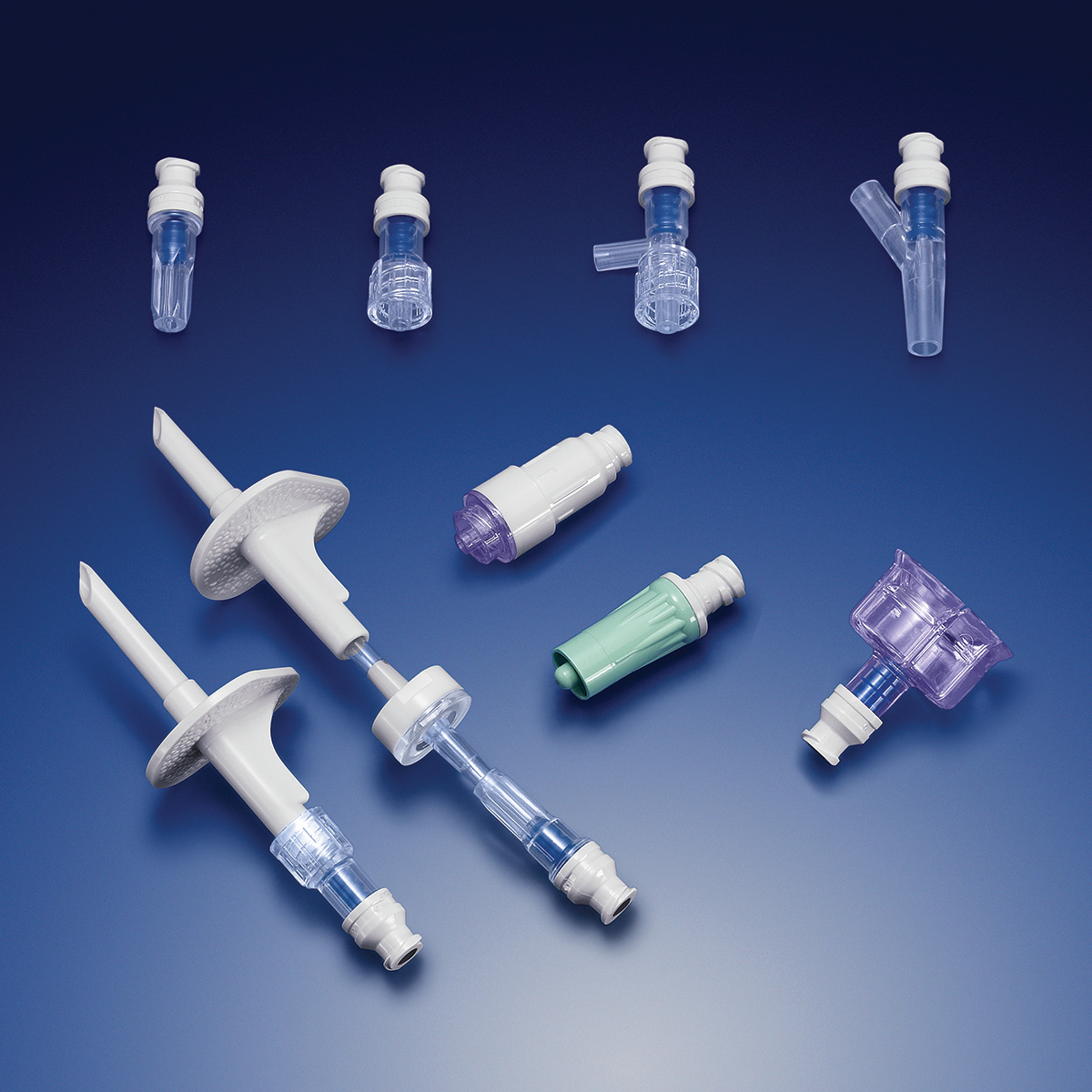
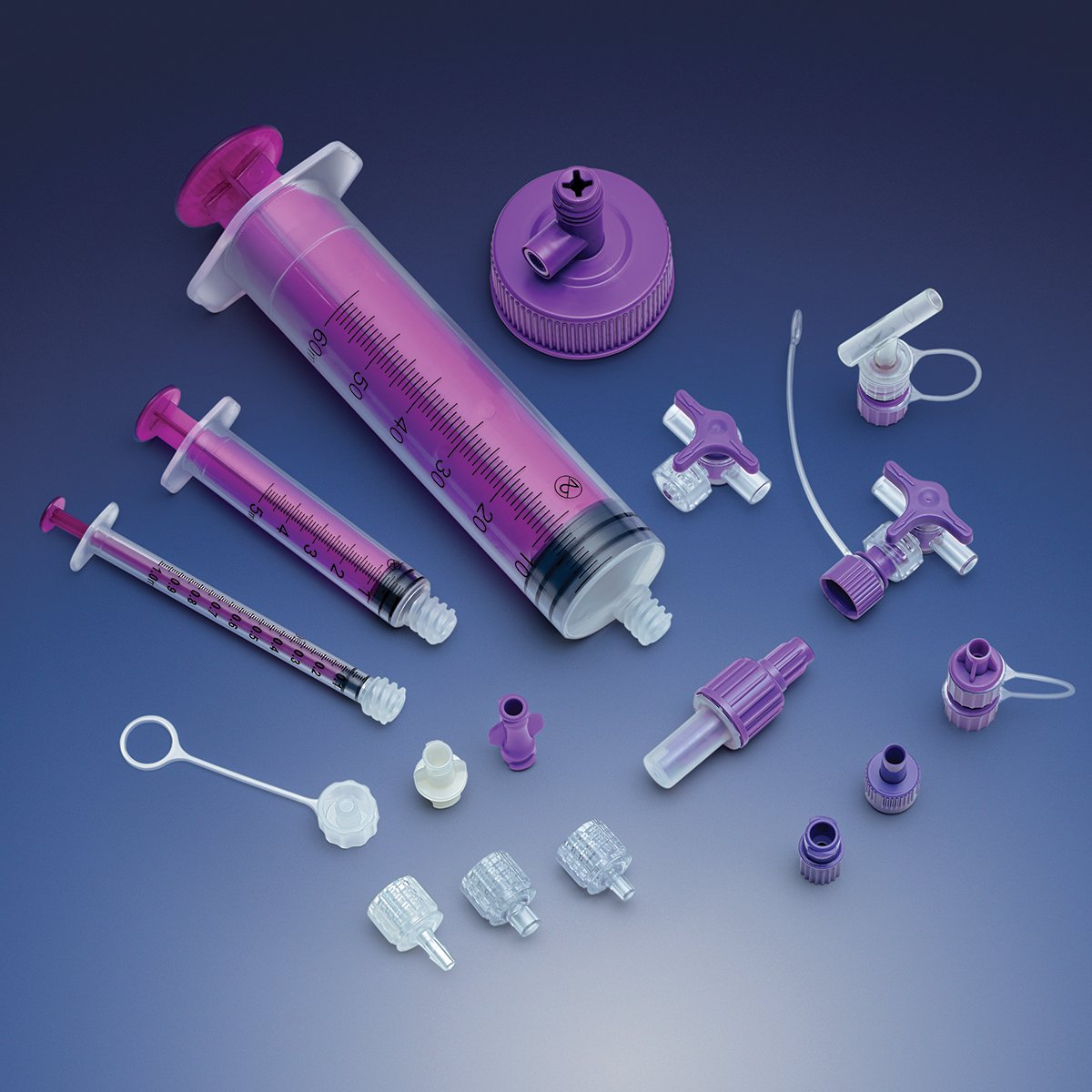
carries a broad product line and supplies components that can be used in a wide
variety of devices, including fluid delivery,
ICU equipment and enteral feeding.
Recently,
the FDA issued a series of guidance documents that provide policies to help
expand the availability of devices to address the COVID-19 public health
emergency. This allows more flexibility for manufacturers to make device
modifications that cover manufacturing limitations or supply shortages related
to COVID-19 without prior approval.
Qosina
is happy to be a second source for components and will help customers find
alternative products for those that are already approved. Customers can browse
qosina.com for critical products to be used in their device manufacturing
projects, and can access material and technical documentation, plus 3D CAD
models for quick product qualification.
Qosina Is a One-Stop Source for COVID-19 Response Components
Qosina
Qosina Is a One-Stop Source for COVID-19 Response Components
Related Products

Enhancing fluid transfer: Qosina adds Pharma+ ™ tubing retainers to product line
QosinaQosina is pleased to announ...

2023 Medical Components Catalog
QosinaThe new 2023 product catalog features thousands of in-stock components depicted in full-scale photos...

High-Flow Check Valves from Qosina
QosinaQosina offers an extensive line of high-flow check valves available in barbed and tubing port co...






%20cropped.jpg)














.jpg)