On February 1 and 2, RAUMEDIC is presenting its newest technologies for the pharmaceutical industry at booth J35 at the Pharmapack in Paris. The polymer specialist focuses on new products for drug delivery, inhalation and injection devices.
January 9, 2017
Helmbrechts –
Drug Delivery
RAUMEDIC develops customized drug delivery systems which offer better handling, exact dosage, and safer application as well as better patient compliance in response to the growing demand for dosing devices that allow an easy and exact drug application.
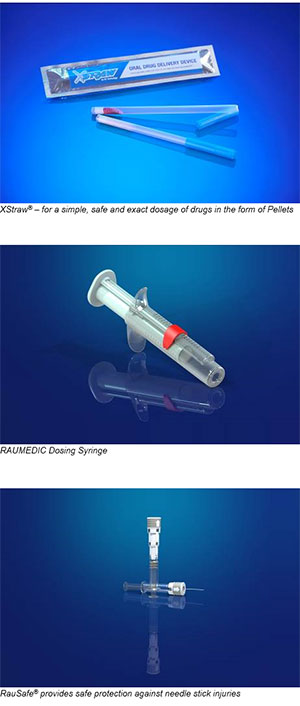
RAUMEDIC and DS TECHNOLOGY have developed a patented system which enables easy and safe dosing of drugs thanks to its multiparticulate dosage forms. In order to prevent inaccurate dosing, the exact pre-dosing is made by the pharmacist. This is one of the reasons why this system is perfectly suited for pediatric and geriatric medicine.
RAUMEDIC is capable to produce and package the plastic components fully automatically. The filling and packaging of XStraw® is handled by our partner DS TECHNOLOGY at its fully automated assembly line.
Another example, which has already been developed by RAUMEDIC together with its customer, is a new dosing syringe with an integrated locking mechanism for the application of liquid drugs. The dosing is adjustable in various steps which permits an individual and exact dosage. Currently available are 0.5 ml, 0.75 ml, 1 ml, 5ml and 10ml versions of the dosing syringe.
Effective Protection against needle stick injuries
Needle stick injuries can result in serious health and financial consequences. Moreover, they are risk factors in doctors' offices, hospitals and in the home care field that have to be taken seriously. To this effect the EU Directive 2010/32/EU for the prevention of needle stick injuries in hospitals and in the health sector came into force in May 2013.
RAUMEDIC’s innovative product RauSafe® offers an innovative safety device for injection systems. It can be adapted to a variety of existing injection systems on the market, provides reliable protection and is simple and intuitive to use. After the injection the active safety device can be simply pushed forward like a telescope. As soon as the needle is completely enclosed, you can hear and feel the system click into the end position.
About RAUMEDIC:
RAUMEDIC AG is a development partner and manufacturer of tubing, molded parts, catheters and components for the medical and pharmaceutical industry. The company processes medical-grade thermoplastic polymers and silicones at a total of four production sites in Germany and in the USA. Furthermore, the medical engineering company develops and produces high-precision pressure measurement systems with microchip technology for the indication areas of neuromonitoring and traumatology.
With 60 years of experience in the fields of extrusion, injection molding and assembly, as well as the commitment of currently 700 employees worldwide, RAUMEDIC is optimally equipped to transform the ideas of its customers into well-engineered product solutions. Customer satisfaction and compliance with all relevant statutory and official requirements are the focus of RAUMEDIC’s conception of quality. The basis of this is a quality management system according to ISO 13485 and 108,000 f² (10,000 m²) of clean room production according to ISO 14644 (class 7).
You can find more information at: www.raumedic.com
View website